SECAUCUS, N.J., Sept. 28, 2017 /PRNewswire/ -- More than 25 percent of patients monitored by lab tests for prescription drug adherence are concurrently taking both opioids and benzodiazepines, according to a new study published online today in the peer reviewed Journal of Addiction Medicine by researchers at Quest Diagnostics (NYSE: DGX), the world's leading provider of diagnostic information services.
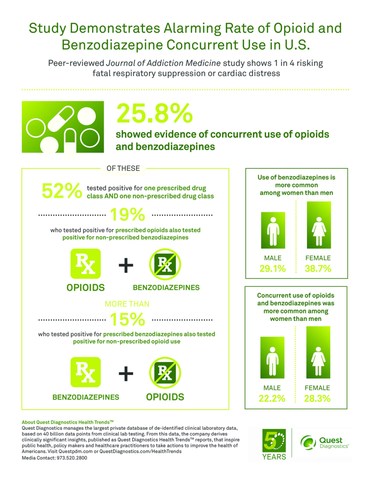
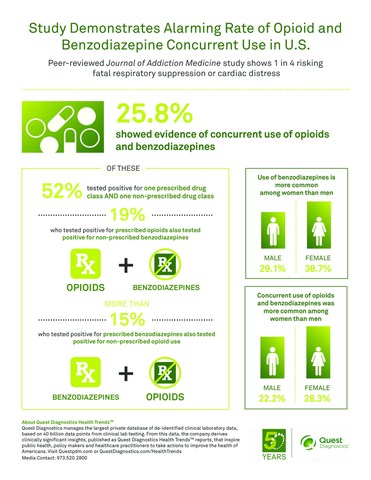
Of results that showed evidence of concurrent use of opioids and benzodiazepines, one in two (52%) included positive results for one prescribed drug and one non-prescribed drug, suggesting drug use without the benefit of clinical oversight. A non-prescribed drug refers to one that was not prescribed for the patient according to the laboratory test order form.
The findings are significant because both opioids and benzodiazepines depress the central nervous system. When combined, respiratory suppression, cardiac distress and even death by overdose can occur.
In August 2016 the United States Food and Drug Administration (FDA) issued a "boxed warning" on both prescription opioids and benzodiazepines alerting prescribers to the dangers of concurrent use. On September 20, 2017, the U.S. Food and Drug Administration emphasized the complexity of the issue, urging against withholding opioid agonist treatments for addiction from patients taking benzodiazepines and noting that "careful medication management by health care professionals" can reduce the risk of serious side effects.
The Quest Diagnostics Health Trends™ study is based on 231,228 sets of test results from 144,535 patients in 48 states tested by Quest Diagnostics between March and December of 2015. The analysis included patients who had been prescribed at least one drug and whose physicians had ordered laboratory drug monitoring.
The study appeared in the Journal of Addiction Medicine, the official peer-reviewed publication of the American Society of Addiction Medicine, and was accompanied by a commentary written by Professor Stefan Kertesz, an addiction medicine specialist at University of Alabama, which noted that the study provides a "valuable window into results of opioid and benzodiazepine assays, as reported from a national laboratory, where physicians and hospitals frequently send specimens for analysis." The study is available here.
The study is believed to be the first national examination of concurrent use of opioids and benzodiazepines that compares objective laboratory data, which provides evidence of recent drug use, to prescription data. Prior analysis of concurrent use of opioids and benzodiazepines has focused on prescription data, but such analysis may not capture situations where patients do not use their drug as prescribed or use other non-prescribed drugs.
"Our findings far exceed previous estimates of combining opiates and benzodiazepines based on prescribing databases alone, suggesting existing prescription databases and monitoring programs do not fully reflect the extent to which individuals may combine these drug classes in the United States," said lead author F. Leland McClure, PhD, MSci, F-ABFT, director, medical science liaison, medical affairs, Quest Diagnostics. "Physicians should be aware of potentially dangerous drug interactions beyond the prescription level, and our data demonstrate these interactions are happening with alarming frequency."
While physicians may prescribe opioids and benzodiazepines together for some patients, doing so carries significant health risks. According to the Centers for Disease Control and Prevention, nearly half of the 52,404 drug overdose deaths in 2015 involved prescription medications. More than 30 percent of opioid-related deaths also involved benzodiazepines.
Novel findings on specific patterns of non-prescribed opioid/benzodiazepine combination
Positivity for prescribed benzodiazepines and prescribed opioids was found in 11.2 percent of all test results. This is similar to the 9.6 percent reported in an analysis of millions of patients filling opioid and benzodiazepine prescriptions. However, this sharply contrasts with the 25.8 percent of all test results that were positive for any type of concurrent use, such as use of a prescribed opioid and a non-prescribed benzodiazepine.
Among other key findings:
- Concurrent use of opioids and benzodiazepines was significantly more common among women (28.3%) than men (22.2%).
- Use of opioids, benzodiazepines, and concurrent use of both drug classes increased significantly with age, but leveled off among those 65 years of age or older. Patients in pain clinics were also more likely to show signs of concurrent use than those receiving primary or other care.
- Medicare patients had significantly higher levels of opioid use, benzodiazepine use, and concurrent use than Medicaid patients and those with commercial insurance or private payer status.
The researchers also found that a large number of test results demonstrating concurrent use tested positive for multiple drugs within a drug group. This was particularly true for opioids.
"These patients are very vulnerable to the dangers of accidental drug overdose from misuse and drug combining. It seems clear that concurrent prescribing is only a part of the concurrent use problem facing the United States," added Dr. McClure. "Opioid prescribing guidelines issued by the CDC advocate for drug testing patients prescribed opioids and limiting benzodiazepines to ensure they are not taking both drugs simultaneously. Careful vigilance by prescribing physicians may help to offset some of the risk posed by patients who may be misusing these substances and other prescription medications."
The strengths of the study include its large scale with national representation of sex, age, and geographic location. Limitations include the amount of detail in subpopulations, potential for selection bias by physicians and the inability of the researchers to verify physician prescribing information.
Quest Diagnostics is one of the largest providers of prescription drug monitoring services in the United States. Services include testing for commonly prescribed drugs, including opioids and other pain medications, central nervous system depressant medications, and certain illicit drugs, such as marijuana (which is illegal in most states), cocaine, and heroin. The company is also a leader in workplace drug testing.
The present analysis is based on a subset of 3.4 million de-identified test results from Quest's 2017 Prescription Drug Misuse in America report, which is available online at www.QuestPDM.com.
About Quest Diagnostics Health Trends
Quest Diagnostics Health Trends™ is a series of reports from Quest Diagnostics on health conditions affecting a large number of Americans. Published in peer-reviewed journals, at scientific conferences and widely as a public service, the reports are designed to inform health care professionals, patients and policymakers about the status of the nation's health. Quest Diagnostics annually issues Prescription Drug Monitoring reports based on data from the Quest Diagnostics Health Trends database, which examines patterns and trends observed from prescription medication monitoring test results. These reports complement the company's Drug Testing Index an analysis of workplace drug testing data.
About Quest Diagnostics
Quest Diagnostics empowers people to take action to improve health outcomes. Derived from the world's largest database of clinical lab results, our diagnostic insights reveal new avenues to identify and treat disease, inspire healthy behaviors and improve health care management. Quest annually serves one in three adult Americans and half the physicians and hospitals in the United States, and our 43,000 employees understand that, in the right hands and with the right context, our diagnostic insights can inspire actions that transform lives. www.QuestDiagnostics.com.
50th Anniversary: In 2017, Quest Diagnostics celebrates 50 years of life-changing results. To learn about our legacy of accomplishments and quest to improve healthcare in the future, visit QuestDiagnostics.com/50Years.
Quest, Quest Diagnostics, and all associated Quest Diagnostics registered or unregistered trademarks are the property of Quest Diagnostics. All third-party marks are the property of their respective owners.

SOURCE Quest Diagnostics